 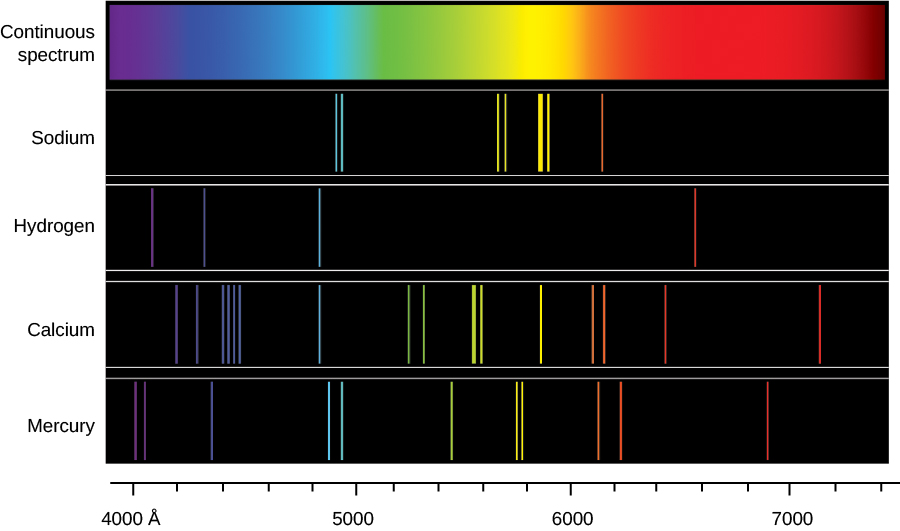
What is the difference in energy between the two energy levels involved in the. Symmetrical multiplets P - P, D - D, etc. Sodium atoms emit a spectral line with a wavelength in the yellow, 589.6mm. (In fact, due to fine structure splitting of the p state, which has not been dealt with here, this line is actually a doublet, i.e. This wavelength corresponds to transitions in atomic sodium in which the valence-electron transitions from a 3p to 3s electronic state. v Line codes indicate additional descriptive information about the appearance of the spectral line. In either case the in-tensity cannot be assumed to be entirely due to the transition indicated in the classication. Relative Strengths for Lines of Multiplets in LS Coupling Normal multiplets S - P, P - D, D - F, etc. The sodium spectrum is dominated by a line of 589 nm wavelength, coming from the transition from the 3p state to the 3s state. Sodium vapor in the upper layers of the sun creates a dark line in the emitted spectrum of electromagnetic radiation by absorbing visible light in a band of wavelengths around 589.5 nm. measured spectral line either is blended with another line or has two identications. A discussion of their normalization as well as more extensive tables are given in Ref. . The total multiplet strengths S M are also listed in this table. Passing white light through sodium vapour produces an absorption spectrum that has a narrow black line at exactly the same place in the yellow. The sodium spectrum is dominated by the bright doublet known as the Sodium D-lines at 588.9950 and 589.5924 nanometers. The satellite lines y n and z n are usually weaker and deviate more from the LS values than the stronger diagonal lines when departures from LS coupling are encountered. Line spectra: The ‘single line’ spectrum of a salted flame can come as a surprise: instead of a broad band of yellow, students see a very narrow band of pure yellow, an emission spectrum. Their strengths normally diminish along the diagonal. The strongest, or principal, lines are situated along the main diagonal of the table and are called x 1, x 2, etc. 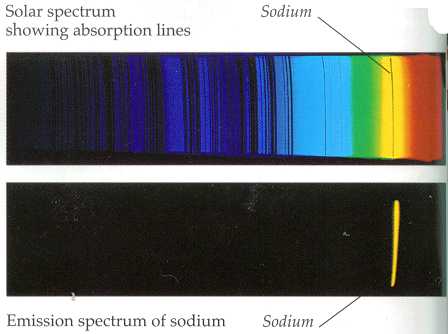
The line at 589.0 has twice the intensity of the line at 589.6 nm. 
From the energy level diagram it can be seen that these lines are emitted in a transition from the 3p to the 3s levels. This table lists relative line strengths for frequently encountered symmetrical (P → P, D → D) and normal (S → P, P → D) multiplets in LS coupling. The sodium spectrum is dominated by the bright doublet known as the Sodium D-lines at 588.9950 and 589.5924 nanometers. Relative Strengths for Lines of Multiplets in LS Coupling And n is the refractive index of standard air. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
